By Lauran Neergaard and Matthew Perrone
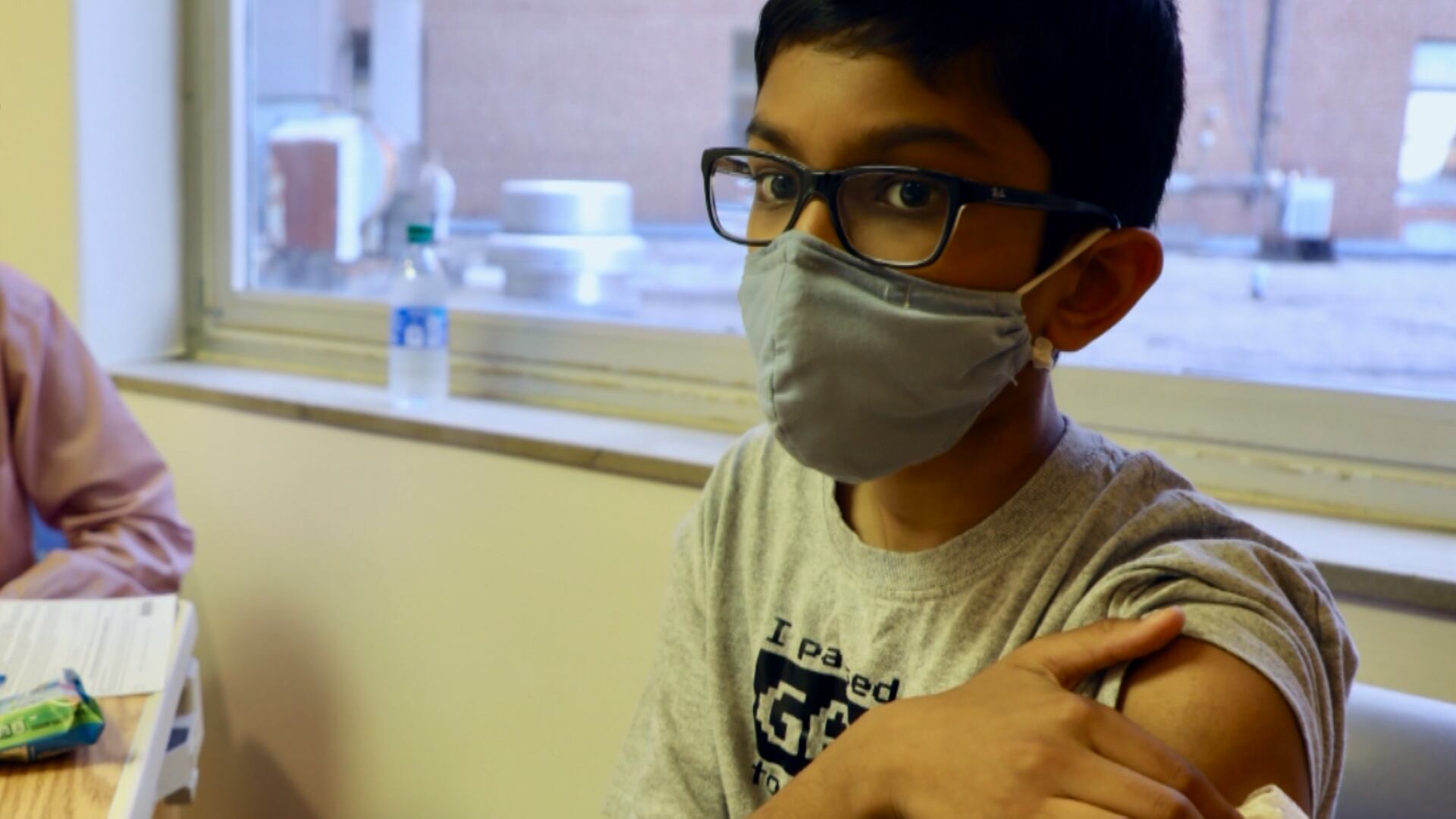
The U.S. gave full approval to Pfizer's COVID-19 vaccine Monday, potentially boosting public confidence in the shots and instantly opening the way for more universities, companies and local governments to make vaccinations mandatory.
The Pentagon promptly announced it will press ahead with plans to require members of the military to get vaccinated amid the battle against the extra-contagious delta variant. Louisiana State University likewise said it will demand its students get the shot.
More than 200 million Pfizer doses have been administered in the U.S. under special emergency provisions — and hundreds of millions more worldwide — since December. In going a step further and granting full approval, the Food and Drug Administration cited months of real-world evidence that serious side effects are extremely rare.
President Joe Biden said that for those who hesitated to get the vaccine until it received what he dubbed the “gold standard” of FDA approval, “the moment you’ve been waiting for is here.”
“Please get vaccinated today,” he said.
Pfizer CEO Albert Bourla called the FDA's action “an important milestone that I think will unlock some of the more skeptical minds.”
Pfizer said the U.S. is the first country to grant full approval of its vaccine, in a process that required a 360,000-page application and rigorous inspections. Never before has the FDA has so much evidence to judge a shot's safety.
The formula, jointly developed with Germany's BioNTech, will be marketed under the brand name Comirnaty.
Moderna has also applied to the FDA for full approval of its vaccine. Johnson & Johnson, maker of the third option in the U.S., said it hopes to do so later this year.
Just over half of the U.S. population is fully vaccinated. Vaccinations in this country bottomed out in July at an average of about a half-million shots per day, down from a peak of 3.4 million a day in mid-April. As the delta variant fills hospital beds, shots are on the rise again, with a million a day given Thursday, Friday and Saturday.
Full approval of Pfizer's COVID-19 vaccine means it meets the same “very high standards required of all the approved vaccines we rely on every day,” said Dr. Jesse Goodman of Georgetown University, a former FDA vaccine chief. That should help “anyone who still has concerns gain confidence” in the shots.
Earlier this month, Defense Secretary Lloyd Austin said he would seek the president’s OK to make the vaccine mandatory by mid-September or once the FDA grants final approval, whichever comes first. On Monday, after the FDA acted, the Pentagon said guidance on vaccinations will be worked out and a timeline will be provided in the coming days.
The approval also opened the way for swift action by colleges to require vaccines and solidified the legal ground for hundreds of universities that have already issued mandates for students and staff.
LSU, with over 30,000 students, acted amid a surge in Louisiana that has repeatedly broken records for the number of people hospitalized with COVID-19. Some major college systems such as the University of Minnesota said they were waiting for FDA approval before making vaccinations mandatory. But some states forbid universities to require shots, including Texas and Florida.
“Mandating becomes much easier when you have full approval,” said Dr. Carlos del Rio of Emory University. “I think a lot of businesses have been waiting for it.”
On the same day the FDA decision came down, New York City announced that all public school teachers and other staffers will have to get vaccinated.
Earlier this month, New York City, New Orleans and San Francisco all imposed proof-of-vaccination requirements at restaurants, bars and other indoor venues. At the federal level, Biden is requiring government workers to sign forms attesting that they have been vaccinated or else submit to regular testing and other requirements.
Anxious Americans increasingly are on board: Close to 6 in 10 favor requiring people to be fully vaccinated to fly or attend crowded public events, according to a recent poll by The Associated Press-NORC Center for Public Affairs Research.
The delta variant has sent cases, deaths and hospitalizations soaring in recent weeks in the U.S., erasing months of progress. Deaths are running at about 1,000 a day on average for the first time since mid-March, and new cases are averaging 147,000 a day, a level last seen at the end of January.
“For weeks we have watched cases go up at an alarming pace among individuals who are not vaccinated while the vaccinated are largely protected,” said Dr. Tomas J. Aragon, director of California’s public health department. “If you are not vaccinated, let this be the milestone that gets you there.”
The FDA, like regulators in Europe and much of the rest of the world, initially allowed emergency use of Pfizer’s vaccine based on a study that tracked 44,000 people 16 and older for at least two months — the time period when serious side effects typically arise.
That’s shorter than the six months of safety data normally required for full approval. So Pfizer kept that study going, and the FDA also examined real-world safety evidence.
Pfizer’s shot will continue to be dispensed to 12- to 15-year-olds under an emergency use authorization, until the company files its its application for full approval.
Normally, doctors can prescribe FDA-approved products for other reasons than their original use. But FDA's acting Commissioner Dr. Janet Woodcock strongly warned that the Pfizer vaccine should not be used “off-label” for children under 12 — a warning echoed by the American Academy of Pediatrics.
Both Pfizer and Moderna have vaccine studies underway in youngsters, and they are using lower doses than those available for people 12 and older.
Pfizer's Bourla said he expects study results from 5- to 11-year-olds by the end of September, but data for those younger than 5 will take a couple of months.
Also, Woodcock said health providers are offering COVID-19 vaccines under agreements with the government that should preclude using Monday's approval as a pretext for offering booster shots to the general population.
Currently, the FDA has authorized third doses of either Pfizer's or Moderna's vaccine only for certain people with severely weakened immune systems, such as organ transplant recipients. For everyone else, the Biden administration is planning for boosters starting in the fall. But the FDA is evaluating that question separately.
In reaching Monday's decision, the FDA said serious side effects remain very rare, such as chest pain and heart inflammation a few days after the second dose, mostly in young men.
As for effectiveness, six months into Pfizer’s original study, the vaccine remained 97% protective against severe COVID-19. Protection against milder infection waned slightly, from a peak of 96% two months after the second dose to 84% by six months.
Those findings came before the delta variant began spreading, but other data from the Centers for Disease Control and Prevention shows the vaccine is still doing a good job preventing severe disease.
___
Associated Press Reporter Jonathan Lemire contributed to this report.
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Department of Science Education. The AP is solely responsible for all content.
Updated on August 23, 2021, at 3:33 p.m. ET with edits.