President Donald Trump in a press conference on Thursday said the Food and Drug Administration is fast-tracking efforts to approve an antiviral therapy, best known for treating malaria, for use in coronavirus cases.
The drug, hydroxychloroquine, was developed more than a half-century ago and is approved for treating malaria, arthritis, and other ailments. Reports out of China and Italy suggest the drug may help, but there is no hard data yet.
"We're going to be able to make that drug available almost immediately," Trump said.
The FDA is working to conduct a "large, pragmatic clinical trial" quickly to confirm the drug's benefit to coronavirus patients, said Commissioner Dr. Stephen Hahn.
He also tempered the president's optimism about the still-untested solution.
"What's also important is not to provide false hope," he said. "We may have the right drug, but it might not be in the appropriate dosage form right now, and it might do more harm than good."
New antiviral therapies face a shorter and less arduous approval process than vaccines, several of which are under development with 12 to 18-month timelines.
"The therapies are something we could move on much faster potentially," Trump said.
He claimed the administration "slashed red tape" to develop new vaccines and therapies.
The president also held out on confirming whether he would invoke the Defense Production Act, which he signed on Wednesday, to mandate the production of needed medical supplies such as N95 respirators and surgical masks.
"If we find that we need something, then we will do that," he said.
Trump repeated that he was open to the federal government buying equity stakes in the airline, cruise, hospitality, and other industries hurt by the outbreak.
The press conference opened with more controversial language placing the blame squarely on China for the outbreak, despite concerns voiced about potentially exacerbating a backlash against Asian Americans.
"We continue our relentless effort to defeat the Chinese virus," Trump said.
As the US braces for the what's the follow after the end of the Title 43 pandemic-related restrictions, experts discuss the ramifications with Cheddar News.
A man who kept a chokehold around the neck of an agitated fellow passenger in the New York City subway has turned himself in on a manslaughter charge.
New York State Senator John Liu spoke with Cheddar News about Asian-American representation in politics, his bill to make Asian-American history mandatory in schools, and the ongoing hate crimes against his community. State Sen. Liu also discussed the debate over student loan forgiveness and gave his take on artificial intelligence in education.
The Senate Banking Committee held a hearing Thursday on the cannabis bill the Secure and Fair Enforcement (SAFE) Banking — a first step toward what advocates hope will be a full vote on the Senate Floor.
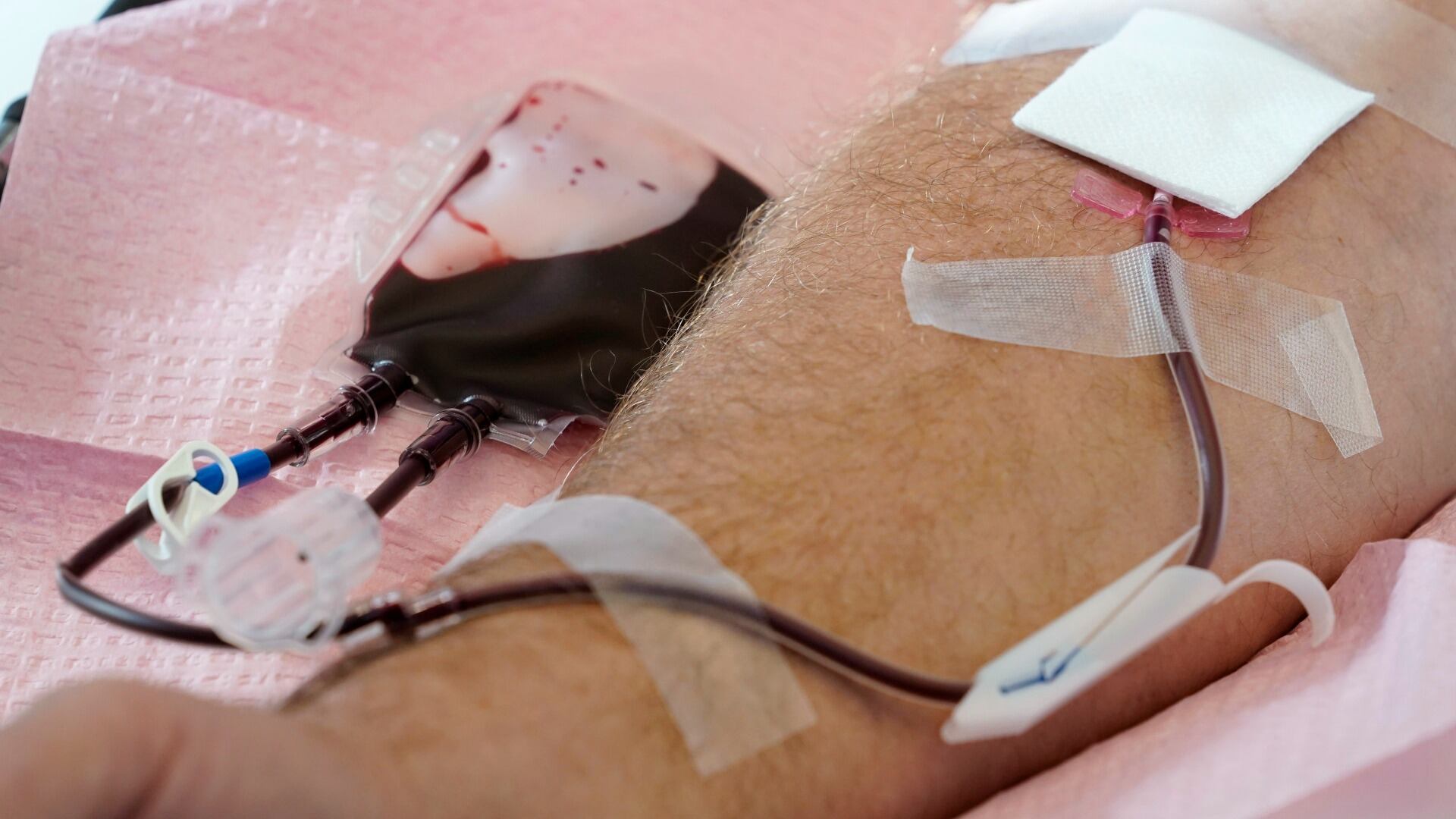
New blood donations rules will allow sexually active gay and bisexual men in monogamous relationships to give in the FDA guidelines ease decades-old restrictions put in place to protect the blood supply from HIV.
During a contentious CNN town hall Wednesday night, former President Donald Trump dug in on his lies about the 2020 election, downplayed the violence on Jan. 6, 2021, and repeatedly insulted the woman whom a civil jury this week found him liable of sexually abusing and defaming.
Criminal defense attorney Tamara Holder joined Cheddar News to break down the charges that New York Republican Congressman George Santos is facing. "When the feds come after you -- and this is for anyone, a politician or not -- they generally have already built their case before they indict you," Holder said, regarding the timeline of Rep. Santos' arrest.
Officials in Missouri's largest city are moving to declare it a sanctuary for people seeking or providing gender-affirming care, defying state officials who are intent on banning it for minors and restricting it for adults.
The Biden administration on Thursday will begin denying asylum to migrants who arrive at the U.S.-Mexico border without first applying online or seeking protection in a country they passed through.
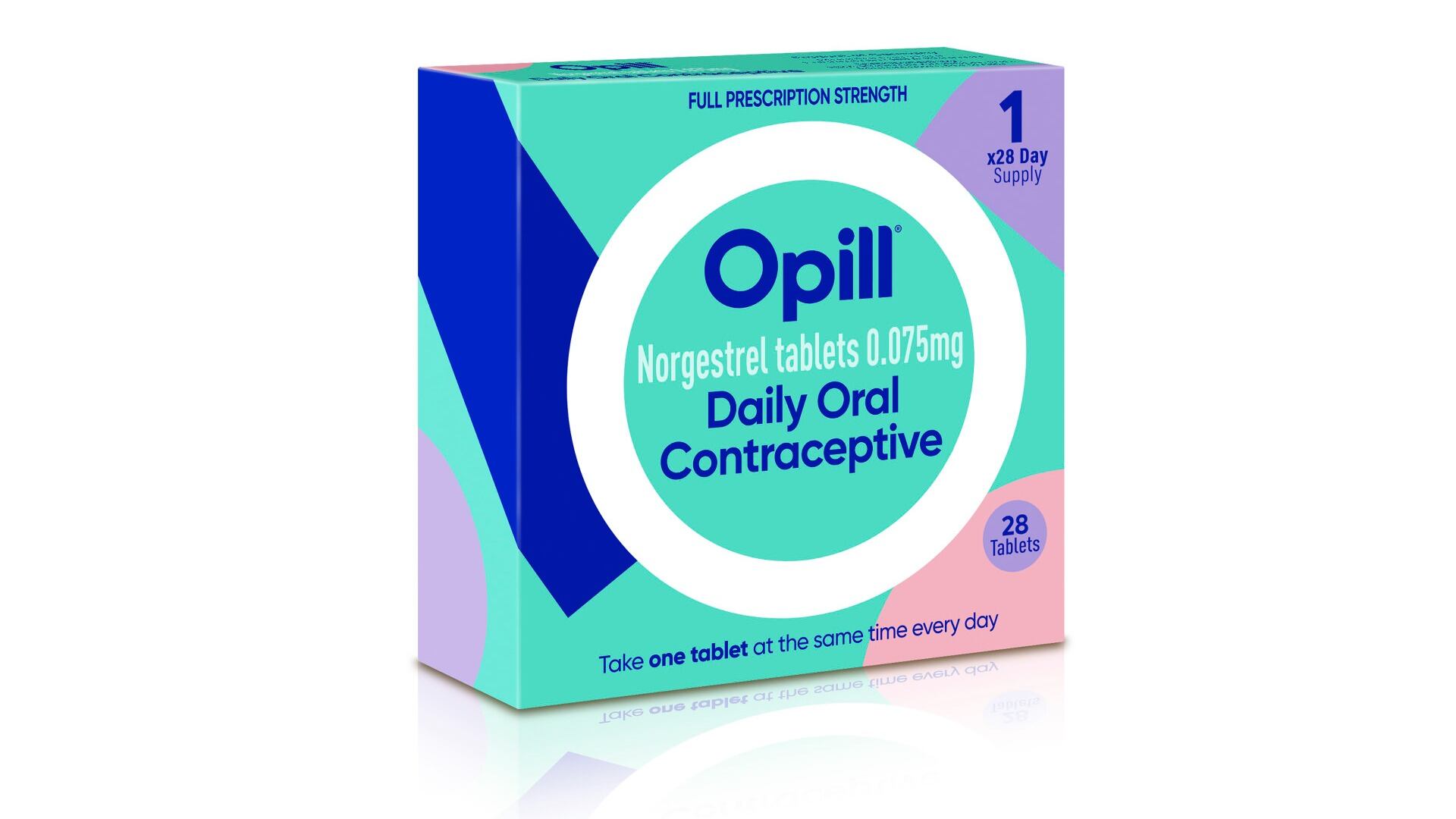
Federal health advisers said Wednesday that a decades-old birth control pill should be sold without a prescription, paving the way for a likely U.S. approval of the first over-the-counter contraceptive medication.