The Food and Drug Administration has revealed that the maker of a brand of eye drops linked to an outbreak of fatal bacterial infections failed to follow multiple safety protocols.
The FDA visited a Global Pharma Healthcare facility in India for an inspection in mid-February and found dozens of issues ranging from dirty equipment and clothing to missing safeguards and procedures.
The inspection came two-and-a-half weeks after the company voluntarily recalled the EzriCare artificial tears product because of possible contamination.
When the recall was issued, there were already 55 reports of adverse events, including eye infections, permanent loss of vision, and at least one death from a bloodstream infection.
As the new year approaches, many people are looking to find a job with a higher salary or are planning to ask their current boss for a raise. Taliya Bashani, real estate attorney and negotiation expert, joined Cheddar News to provide tips on how to properly and better negotiate better financial terms.
Meta says it will start testing a program that would allow posts from Threads to appear on other social media sites.
Several healthcare companies are reportedly joining President Biden's artificial intelligence risk management plan.
The Biden administration says it will impose inflation penalties on dozens of drug makers to lower costs for those on Medicare.
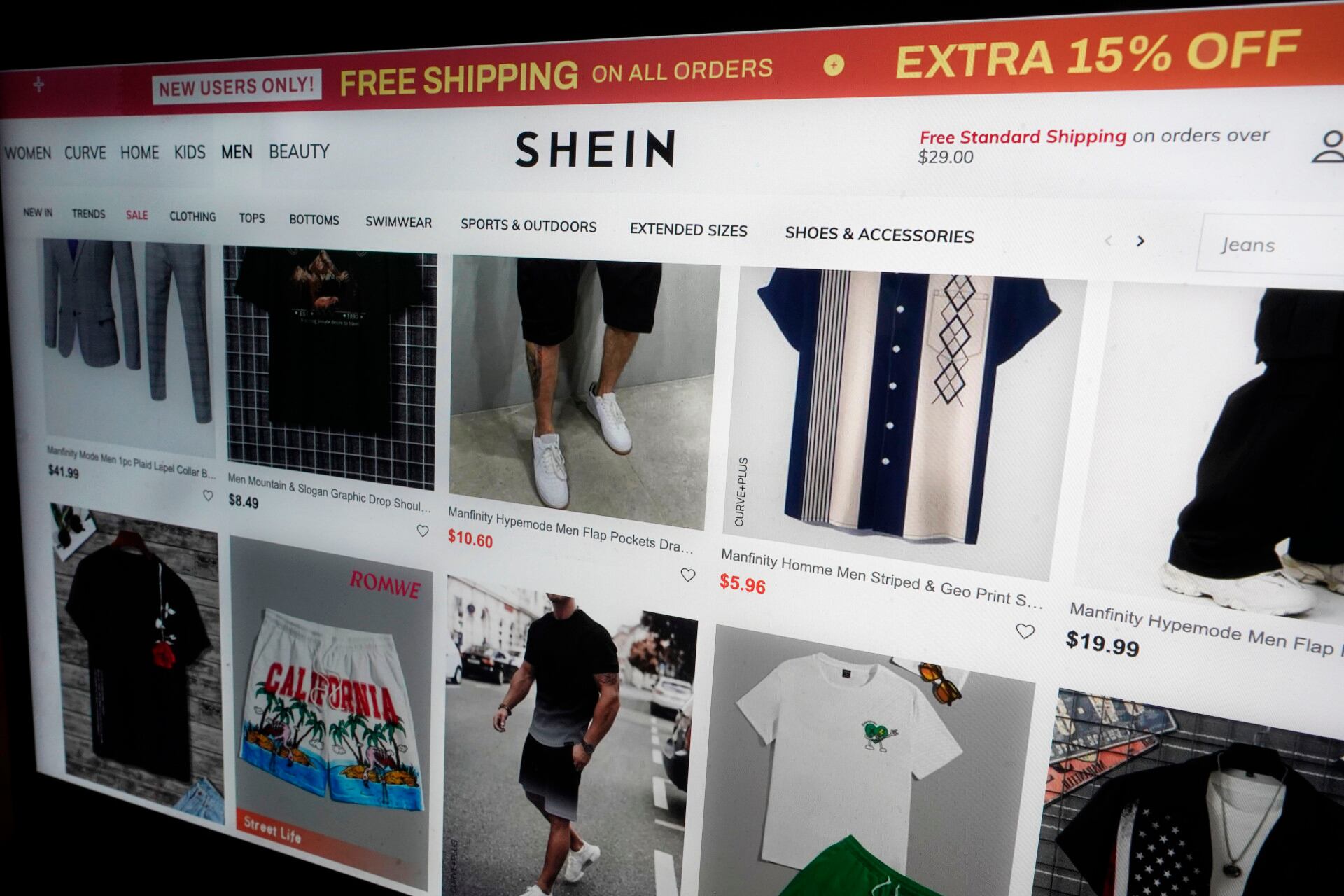
Two of the top low-cost online retailers are going head to head in a new legal battle. Cheddar News' Michelle Castillo breaks the lawsuit down.
The number of Americans filing for jobless benefits fell last week as the labor market continues to thrive despite high interest rates and elevated costs.
The earliest version of Disney's Mickey Mouse will become public domain on Jan. 1, 2024.
The toy magic oven called the Cookeez Makery is one of the hottest toys for kids this holiday season.
In the UK, IKEA is looking to give away some meatballs, and not just regular-size meatballs.
Dwayne 'The Rock' Johnson is set to star in a movie playing MMA and UFC legend, Mark Care, while Kevin Hart will headline a boxing project over on Peacock.